Methodology for Hips
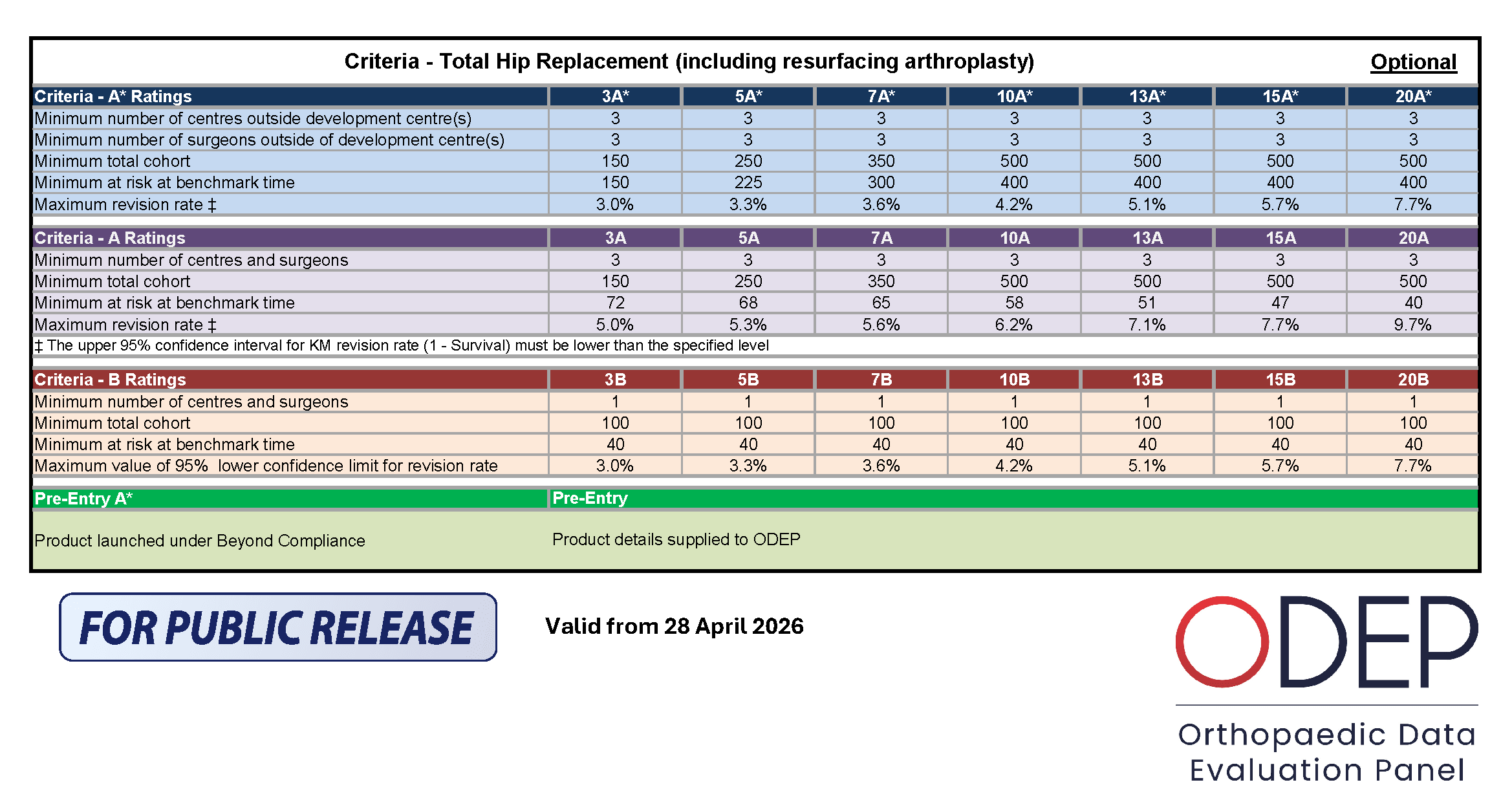
Total hip replacement is a major operation which involves replacing the native hip joint, either by a standard stemmed device or a resurfacing arthroplasty. In both cases the acetabulum is removed and replaced by an implant. In the standard THR the head of the femur is excised and a stem passed down the inside of the proximal femur on to which a head is attached via a taper / trunnion system. In a resurfacing procedure the femoral head is left in situ and it is resurfaced. Fixation of all these implants is either by using a bone cement (“Cemented”) or by their bone facing surface attracting and becoming incorporated in the local host bone (“Uncemented”).
Although the ODEP is broadly the same system for assessing and benchmarking TJRs, there are differences in the approach which have been developed to match the differences between the joints that are being benchmarked.
In the case of THRs, ODEP assess and benchmark stems and acetabular components separately. The reasons for this are as follows:
1. Some manufacturers, albeit a small number, only manufacture either cups or stems
2. ODEP does not consider it possible to assess and benchmark all the constructs that are used in the UK, let alone around the world because it is an enormous number. Even within a brand there is often several combinations offered by the manufacturer and there continues to be a considerable amount of mixing and matching of femoral and acetabular components from more than one manufacturer.
In the case of surface replacements the same standards will apply.
However, as with all TJRs there are several stipulations that must be met:
• Firstly, the implant must be manufactured by the said manufacturer unless clearly stated otherwise.
• The manufacturing process and the same standards are expected to be followed for the whole of the brand being submitted.
For standard stems, (not patient matched)
• The material composition is identical across the range
• Cemented and uncemented brands are assessed separately and require separate submissions
• The implant surface finish is identical across the range
• The taper/trunnion design is identical across the range
• Implant specifications are identical across the range, other than to accommodate increases in size of the manufacturer’s “Standard” stem width and offset
• Implants marketed as being “Short stem” implants will be considered as a separate group
• For modular necks, only one submission will be required if the metal from which the modular neck is manufactured is the same as the stem. If a different metal is used in the manufacture of the neck a separate submission will be required
• At present, the head size recommended by the manufacturer to be used with this stem should be of a standard size (28, 32 or 36mm) and can be made of the standard materials used for the manufacture of standard modular femoral heads.
Rating for standard Acetabular components
It is used as a primary hip replacement
• The material from which it is made is identical across the range
• The bone facing surface finish/coating is identical across the range
• The implant specifications are the same across the range (including modular liner locking mechanism), other than to accommodate incremental increases in cup size and screw holes.
• The cups / liners should accommodate standard head sizes (28, 32 or 36 mm).
Bundling and Camouflage – Should brands be increasingly split up?
It is accepted that some options marketed within in a brand may not be used in sufficient numbers to qualify for a separate rating. It is not ODEP’s intention to exclude these important options from the ODEP process.
However with more data available to manufacturers to be able to assess the performance of different versions of each of their brands, (leading to increased confidence for patients and their surgeons), ODEP see advantages in manufacturers submitting separate data around certain attributes with a brand. The situation is under review (2021).
With femoral components the following are examples of attributes that could be split
• “Long Stem” options within a brand when not marketed separately
• Collared and collarless versions of a femoral stems that are otherwise identical
• Variations in “Offset”
With Acetabular components the following could be split
• Peripheral expanded shells should be analysed separately to hemispherical shells
• Ceramic and polyethylene liners with modular cementless systems should be considered as separate implants and require separate submissions
• Modular dual mobility components and constrained liners/cups should be considered as separate implants with an individual rating and require separate submissions. Separate submissions will be required for cemented and uncemented dual mobility cups.
• We expect that differences in liner design, such as lateralising, posterior build-up and face changing options would be included within one rating if the material composition and locking mechanism is identical.
Please see the Rules for bundling and the avoidance of camouflage on this website.
With regard to the above, it is likely that bundling will be allowed in certain circumstances as with other joints. Different implants (i.e. long vs standard stems) within families will be able to be awarded the same rating based on the larger numbers provided by the standard version if both versions are demonstrably identical on survival analysis at the time point satisfied by the ODEP rating requested. As with other TJRs, the options in the bundle would be expected to have been available on the market for the same length of time.
Patient Matched Implants
Reference is made under the heading “ODEP methodology” to this relatively new group of implants that are made for individual patients on the basis of a scan (CT or MRI) of the patient’s joint. They may be made from bar or by Additive surface layer technology, sometimes referred to as 3D printing.
ODEP will benchmark these implants provided every attribute within the implant envelope is the same. That is to say that all the requirements that are mentioned above are the same (material, bone facing surface, trunnion etc). The only differences that would be permitted are changes in shape / morphology that are embraced by the primary purpose of this type of implant.
Data Sources
Please refer to the fuller section on Data sources by opening the appropriate Tab.
ODEP expects that data sources used by manufacturers for their submissions should come from validate sources. Registry data will always be accepted from registries which comply with the ISAR standards for a level 1 registry where compliance and completeness are over 90%. Peer reviewed publications and presentations are accepted provided they cover the areas of inquiry stipulated in the submission form.
ODEP encourages manufacturers to monitor their products carefully and in-house data that complies with ISO 14155:2020 standards and is validated (or can be validated by ODEP) is accepted in certain circumstances. The data collection must fit the submission form perfectly. If in house data is submitted from hospitals in an area of the world where there is a registry, data from that registry (however limited) must be included.

